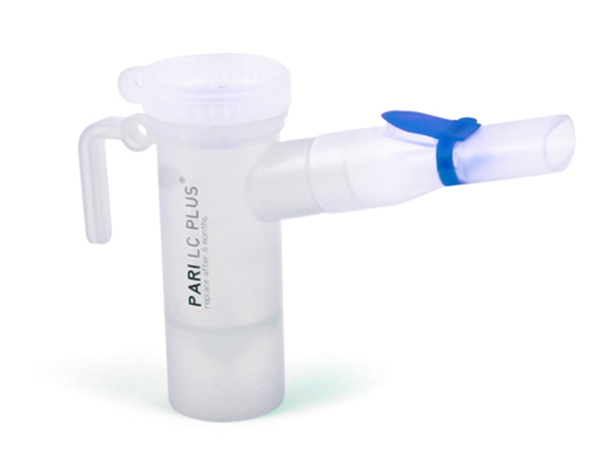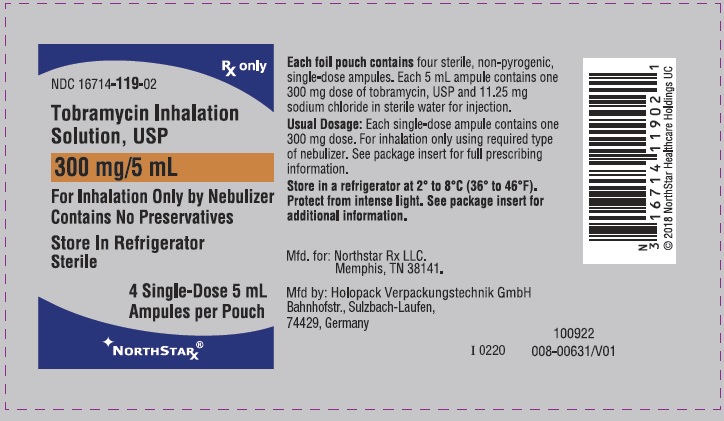
These highlights do not include all the information needed to use TOBRAMYCIN INHALATION SOLUTION safely and effectively. See full prescribing information for TOBRAMYCIN INHALATION SOLUTION. TOBRAMYCIN INHALATION solution, for oral inhalation use
Tobramycin In 0.225 % Sodium Chloride Inhalation: Uses, Side Effects, Interactions, Pictures, Warnings & Dosing - WebMD

Tobramycin by Amneal Pharmaceuticals LLC / Amneal Pharmaceuticals, LLC / Catalent Pharma Solutions, LLC TOBRAMYCIN solution